Food recall market attracts insurers
Reprints
Food-related contamination insurance coverage is an outlier in the hard commercial property/casualty market, with policyholders continuing to obtain rate decreases.
But there is concern over the recent recalls of contaminated peanut butter and baby formula, which along with other cyclical market forces, could lead to a hardening market within the next year or so, experts say.
They point out also that a return to a more normal pace of federal plant inspections, which slowed during the pandemic, is expected to lead to an increase in claims.
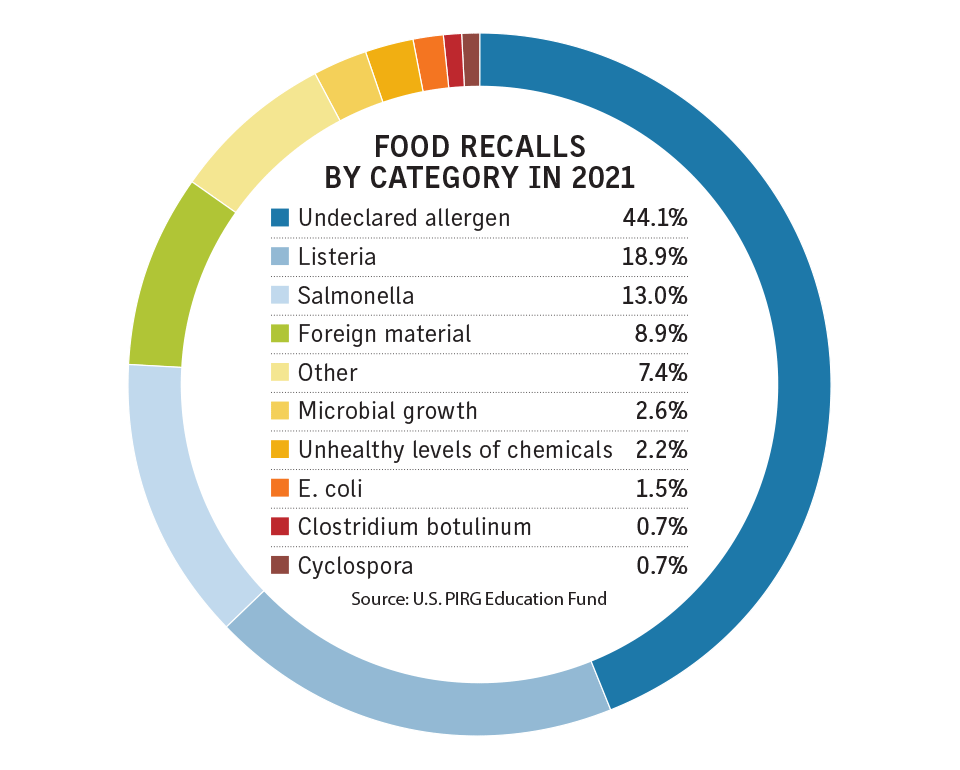
Experts say food recall coverage, which is generally written under policies labeled contaminated products or similar nomenclature, is written by around 30 different markets split about equally between the United States and London. It is generally written in the excess and surplus lines market.
A significant portion of the business is written by managing general agencies, which are often funded by noninsurance sources, although many traditional insurers are active in the segment as well.
Food contamination issues are generally overseen by the U.S. Food and Drug Administration and, to a lesser extent, the Department of Agriculture.
Experts say a major factor in the market has been the FDA Food Safety Modernization Act. The measure, which was signed into law by President Barack Obama in January 2011, shifted the focus from responding to contamination to preventing it.
Experts say the most common claims relate to allergens — including nuts, eggs, gluten and sesame seeds — that purportedly are not listed as product ingredients.
Other claims are related to pathogens, such as E. coli and listeria, with foreign materials making up the third category.
Observers say insurance claims related to recalls are infrequently litigated.
Capacity influx
Dru Wilson, Tampa, Florida-based national manufacturing and distribution vertical leader for USI Insurance Services Inc., said the food recall insurance market is in a slightly better state than it has been for many years.
Michael Capleton, London-based underwriter, product recall, specialty lines, at CFC Underwriting Ltd., described the market as “quite soft.”
“We have probably as much capacity now in the marketplace as ever before,” said Steve Kluting, Grand Rapids, Michigan-based national director, product recall, food, beverage & agribusiness practice group, at Arthur J. Gallagher & Co.
“We’ve got more insurers, more capacity, broader forms,” said Geoffrey Mills, Tampa-based product recall practice leader for Marsh LLC.
Observers say there is $300 million to $600 million in capacity available and that rate reductions are running at about 5% to 10%.
“People are not putting up $25 million in limits right and left,” said Sommer Chanady, Newport Beach, California-based senior vice president at Hub International Ltd. It depends on the risk, its place in the supply chain, and what is involved, she said.
Rob Balogh, Chicago-based executive vice president of product recall for Amwins, attributed the market’s competitiveness to the large increase in new capacity funded by MGAs and outside capital.
As in other areas of insurance, “these underwriters want to build something from the ground up and then sell it,” he said. MGAs “have the ability to do things standard markets cannot, and because of that it’s caused severe competitiveness in the marketplace,” Mr. Balogh said.
The pandemic may also be affecting the market, observers say.
According to the Denver-based United States Public Interest Research Group Education Fund, the USDA Food Safety and Inspection Service and the FDA reported 270 food and beverage recalls in 2021, the lowest number since 2017, when they totaled 239. The public interest organization said the decline could be attributable, in part, to agency staffing shortages during the pandemic.
Shawn McCleary, Chicago-based recall team associate director for Willis Towers Watson PLC, said he expects a post-pandemic increase in food contamination claims, as postponed plant inspections resume.
“Now that things are slightly back to normal, we’re going to see more FDA site visits, and, usually when that happens, we see more tests for biological contaminants,” he said. “We predict that will be more losses in the industry for the next 12 months.”
“Some folks recognized in hindsight that some corners were cut,” and, anecdotally, “we’re starting to see some of that play out,” although these may not necessarily rise to the level of an insurance recall claim, Mr. Kluting said.
Many observers say the soft market is likely for at least another year or two, assuming there is not another major incident along the lines of the Jif peanut butter recall.
“I continue to see a competitive marketplace at least for the next couple of years,” Mr. Mills said.
Tainted products highlight risks
Peanut butter and baby formula are two recent widely publicized recalls by the U.S. Food and Drug Administration.
The FDA announced in May that along with the Centers for Disease Control and Prevention and state and local partners, it was investigating a multistate outbreak of salmonella infections linked to certain Jif peanut butter products produced at a J.M. Smucker Co. facility in Lexington, Kentucky, and that Smucker had voluntarily recalled the products.
The recall has spread to dozens of other products that use peanut butter as an ingredient.
“It’s not just them, it’s every one of their clients that their product is going to,” said Dru Wilson, national manufacturing and distribution vertical leader for USI Insurance Services Inc. in Tampa, Florida.
In February, the FDA warned consumers not to use powdered infant formula products from Abbott Nutrition’s Sturgis, Michigan, plant, and parent company Abbott Laboratories voluntarily stopped production and began a product recall.
In May, the FDA issued guidance outlining “increased flexibilities” regarding importing infant formula products to increase their availability.
“The peanut butter and infant formula recalls are going to be an awakening to the recall market,” said Natasha McLean, Kansas City, Missouri-based vice president, national risk practice consulting leader for product recall and contamination, for Lockton Cos. LLC.